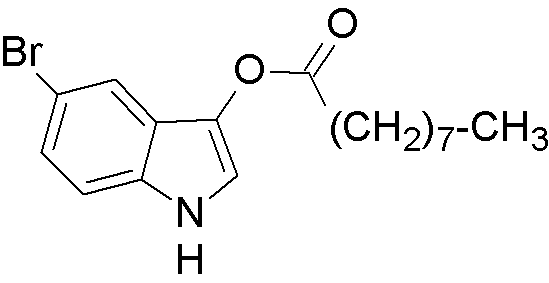
5-Bromo-3-indoxyl nonanoate is widely utilized in research focused on:
- Biochemical Assays: This compound serves as a substrate in various enzymatic assays, allowing researchers to study enzyme activity and kinetics effectively.
- Histochemical Staining: It is employed in histochemistry for visualizing specific cellular components, aiding in the identification of tissue types and disease states.
- Drug Development: The compound is used in the pharmaceutical industry to develop new drugs, particularly those targeting specific biological pathways involving indole derivatives.
- Environmental Monitoring: It can be utilized in environmental science to assess the presence of certain pollutants, contributing to ecological studies and assessments.
- Educational Purposes: This chemical is often included in laboratory courses for students studying organic chemistry and biochemistry, providing hands-on experience with indole derivatives.
General Information
Properties
Safety and Regulations
Applications
5-Bromo-3-indoxyl nonanoate is widely utilized in research focused on:
- Biochemical Assays: This compound serves as a substrate in various enzymatic assays, allowing researchers to study enzyme activity and kinetics effectively.
- Histochemical Staining: It is employed in histochemistry for visualizing specific cellular components, aiding in the identification of tissue types and disease states.
- Drug Development: The compound is used in the pharmaceutical industry to develop new drugs, particularly those targeting specific biological pathways involving indole derivatives.
- Environmental Monitoring: It can be utilized in environmental science to assess the presence of certain pollutants, contributing to ecological studies and assessments.
- Educational Purposes: This chemical is often included in laboratory courses for students studying organic chemistry and biochemistry, providing hands-on experience with indole derivatives.
Documents
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
Product Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
*Catalog Number
*Lot Number
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.
*Catalog Number
*Lot Number
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
DownloadProduct Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
DownloadCertificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
*Catalog Number
*Lot Number
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.