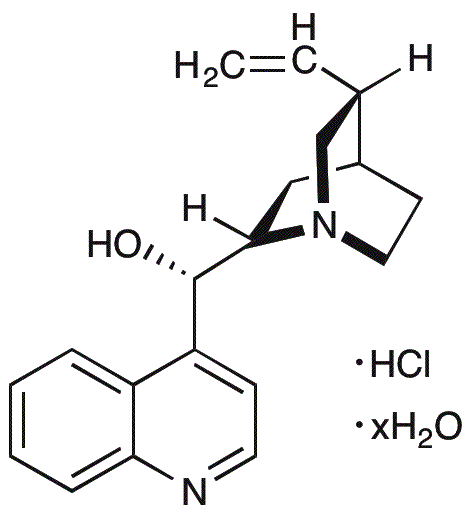
Cinchonine monohydrochloride hydrate is widely utilized in research focused on:
- Pharmaceutical Development: This compound is integral in the synthesis of various medications, particularly in the treatment of malaria and other parasitic infections, enhancing therapeutic efficacy.
- Analytical Chemistry: It serves as a chiral resolving agent in chromatography, helping researchers separate enantiomers effectively, which is crucial for drug formulation and quality control.
- Natural Product Synthesis: Used as a building block in the synthesis of complex natural products, it aids chemists in developing new compounds with potential medicinal properties.
- Biochemical Research: Its role as a ligand in biochemical assays allows for the study of various biological processes, providing insights into enzyme activity and receptor interactions.
- Organic Synthesis: Cinchonine monohydrochloride hydrate is employed in asymmetric synthesis, offering advantages in producing compounds with high optical purity, which is essential in pharmaceuticals.
General Information
Properties
Safety and Regulations
Applications
Cinchonine monohydrochloride hydrate is widely utilized in research focused on:
- Pharmaceutical Development: This compound is integral in the synthesis of various medications, particularly in the treatment of malaria and other parasitic infections, enhancing therapeutic efficacy.
- Analytical Chemistry: It serves as a chiral resolving agent in chromatography, helping researchers separate enantiomers effectively, which is crucial for drug formulation and quality control.
- Natural Product Synthesis: Used as a building block in the synthesis of complex natural products, it aids chemists in developing new compounds with potential medicinal properties.
- Biochemical Research: Its role as a ligand in biochemical assays allows for the study of various biological processes, providing insights into enzyme activity and receptor interactions.
- Organic Synthesis: Cinchonine monohydrochloride hydrate is employed in asymmetric synthesis, offering advantages in producing compounds with high optical purity, which is essential in pharmaceuticals.
Documents
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
Product Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Numéro de catalogue
Numéro de lot/série
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.
Numéro de catalogue
Numéro de lot/série
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
DownloadProduct Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
DownloadCertificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Numéro de catalogue
Numéro de lot/série
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.