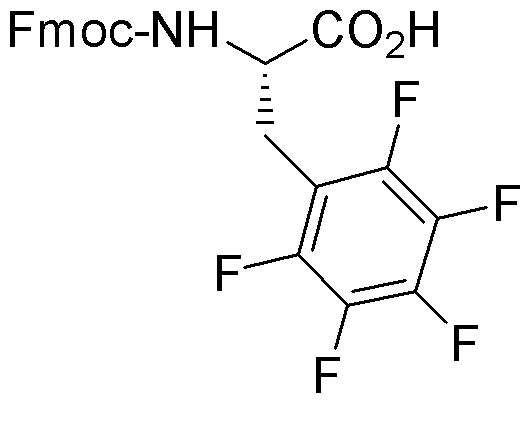
Fmoc-pentafluoro-L-phenylalanine is widely utilized in research focused on:
- Peptide Synthesis: This compound serves as a key building block in the synthesis of peptides, particularly in solid-phase peptide synthesis, allowing for the incorporation of fluorinated amino acids that enhance peptide stability and bioactivity.
- Drug Development: Its unique fluorinated structure is beneficial in medicinal chemistry, where it can improve the pharmacokinetic properties of drug candidates, making them more effective and selective.
- Bioconjugation: Researchers use this compound to create bioconjugates, which are essential in developing targeted therapies and diagnostic agents, particularly in cancer treatment.
- Material Science: The fluorinated properties contribute to the development of advanced materials with improved chemical resistance and thermal stability, useful in coatings and electronics.
- Research on Protein Folding: It aids in studies related to protein folding and stability, providing insights into protein interactions and functions, which is crucial for understanding various biological processes.
General Information
Properties
Safety and Regulations
Applications
Fmoc-pentafluoro-L-phenylalanine is widely utilized in research focused on:
- Peptide Synthesis: This compound serves as a key building block in the synthesis of peptides, particularly in solid-phase peptide synthesis, allowing for the incorporation of fluorinated amino acids that enhance peptide stability and bioactivity.
- Drug Development: Its unique fluorinated structure is beneficial in medicinal chemistry, where it can improve the pharmacokinetic properties of drug candidates, making them more effective and selective.
- Bioconjugation: Researchers use this compound to create bioconjugates, which are essential in developing targeted therapies and diagnostic agents, particularly in cancer treatment.
- Material Science: The fluorinated properties contribute to the development of advanced materials with improved chemical resistance and thermal stability, useful in coatings and electronics.
- Research on Protein Folding: It aids in studies related to protein folding and stability, providing insights into protein interactions and functions, which is crucial for understanding various biological processes.
Documents
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
Product Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Numéro de catalogue
Numéro de lot/série
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.
Numéro de catalogue
Numéro de lot/série
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
DownloadProduct Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
DownloadCertificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Numéro de catalogue
Numéro de lot/série
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.