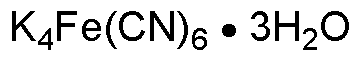Potassium ferrocyanide trihydrate is widely utilized in research focused on:
- Analytical Chemistry: It serves as a reagent in various analytical techniques, including colorimetric assays, to detect and quantify metal ions due to its ability to form stable complexes.
- Food Industry: This compound is used as an anti-caking agent in salt and other food products, improving flowability and preventing clumping, which enhances product quality.
- Photography: It plays a role in the production of photographic chemicals, particularly in the formulation of developers and fixers, contributing to image clarity and stability.
- Electroplating: In the electroplating industry, it acts as a source of cyanide ions, which are essential for the deposition of metals like gold and silver, ensuring a smooth and even coating.
- Biochemistry: Researchers utilize it in various biochemical assays and experiments, particularly in studies involving enzyme activity and metal ion interactions, providing insights into biological processes.
General Information
Properties
Safety and Regulations
Applications
Potassium ferrocyanide trihydrate is widely utilized in research focused on:
- Analytical Chemistry: It serves as a reagent in various analytical techniques, including colorimetric assays, to detect and quantify metal ions due to its ability to form stable complexes.
- Food Industry: This compound is used as an anti-caking agent in salt and other food products, improving flowability and preventing clumping, which enhances product quality.
- Photography: It plays a role in the production of photographic chemicals, particularly in the formulation of developers and fixers, contributing to image clarity and stability.
- Electroplating: In the electroplating industry, it acts as a source of cyanide ions, which are essential for the deposition of metals like gold and silver, ensuring a smooth and even coating.
- Biochemistry: Researchers utilize it in various biochemical assays and experiments, particularly in studies involving enzyme activity and metal ion interactions, providing insights into biological processes.
Documents
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
Product Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Numéro de catalogue
Numéro de lot/série
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.
Numéro de catalogue
Numéro de lot/série
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
DownloadProduct Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
DownloadCertificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Numéro de catalogue
Numéro de lot/série
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.