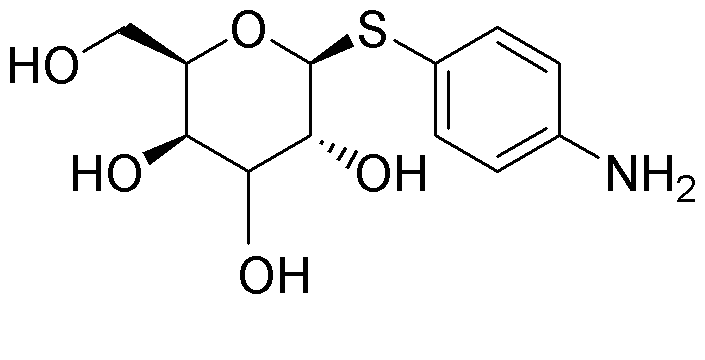
S-p-Aminophenyl-b-D-thiogalactoside is widely utilized in research focused on:
- Biochemical Research: This compound serves as a substrate for studying galactoside transport systems, aiding researchers in understanding cellular uptake mechanisms.
- Drug Development: It is used in the development of novel therapeutics targeting specific glycosylation pathways, which can lead to advancements in treatments for various diseases.
- Enzyme Activity Assays: The compound is employed in assays to evaluate the activity of glycosidases, providing insights into enzyme kinetics and potential inhibitors.
- Cell Biology: It helps in the investigation of cell signaling pathways, particularly those involving glycoproteins, enhancing our knowledge of cellular interactions.
- Diagnostics: S-p-Aminophenyl-b-D-thiogalactoside can be used in diagnostic tests to detect specific enzymes related to certain medical conditions, offering a valuable tool for early diagnosis.
General Information
Properties
Safety and Regulations
Applications
S-p-Aminophenyl-b-D-thiogalactoside is widely utilized in research focused on:
- Biochemical Research: This compound serves as a substrate for studying galactoside transport systems, aiding researchers in understanding cellular uptake mechanisms.
- Drug Development: It is used in the development of novel therapeutics targeting specific glycosylation pathways, which can lead to advancements in treatments for various diseases.
- Enzyme Activity Assays: The compound is employed in assays to evaluate the activity of glycosidases, providing insights into enzyme kinetics and potential inhibitors.
- Cell Biology: It helps in the investigation of cell signaling pathways, particularly those involving glycoproteins, enhancing our knowledge of cellular interactions.
- Diagnostics: S-p-Aminophenyl-b-D-thiogalactoside can be used in diagnostic tests to detect specific enzymes related to certain medical conditions, offering a valuable tool for early diagnosis.
Documents
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
Product Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
*Catalog Number
*Lot Number
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.
*Catalog Number
*Lot Number
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
DownloadProduct Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
DownloadCertificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
*Catalog Number
*Lot Number
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.