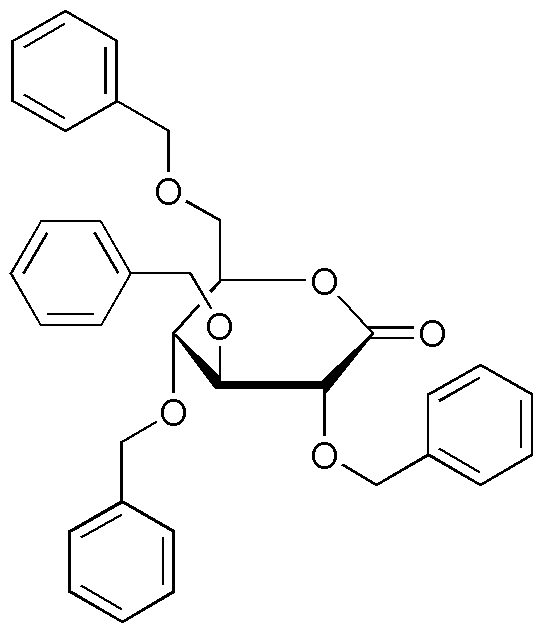
2,3,4,6-tetra-O-benzyl-D-glucono-1,5-lactone is widely utilized in research focused on:
- Synthesis of Glycosides: This compound serves as a versatile building block in the synthesis of glycosides, which are important in pharmaceuticals and biochemistry for their role in drug development and natural product synthesis.
- Protecting Group in Organic Chemistry: It acts as a protecting group for hydroxyl functionalities during multi-step organic synthesis, allowing chemists to selectively modify other parts of the molecule without affecting the protected sites.
- Research in Carbohydrate Chemistry: This lactone is utilized in carbohydrate chemistry studies, helping researchers understand glycosidic bond formation and the reactivity of sugars, which is crucial for developing new carbohydrate-based materials.
- Pharmaceutical Applications: Due to its structural properties, it is explored in the formulation of drug delivery systems, enhancing the solubility and stability of therapeutic agents.
- Food Industry Applications: It can be used in food chemistry for flavoring and as a stabilizing agent, contributing to the development of new food products with enhanced taste and shelf life.
General Information
Properties
Safety and Regulations
Applications
2,3,4,6-tetra-O-benzyl-D-glucono-1,5-lactone is widely utilized in research focused on:
- Synthesis of Glycosides: This compound serves as a versatile building block in the synthesis of glycosides, which are important in pharmaceuticals and biochemistry for their role in drug development and natural product synthesis.
- Protecting Group in Organic Chemistry: It acts as a protecting group for hydroxyl functionalities during multi-step organic synthesis, allowing chemists to selectively modify other parts of the molecule without affecting the protected sites.
- Research in Carbohydrate Chemistry: This lactone is utilized in carbohydrate chemistry studies, helping researchers understand glycosidic bond formation and the reactivity of sugars, which is crucial for developing new carbohydrate-based materials.
- Pharmaceutical Applications: Due to its structural properties, it is explored in the formulation of drug delivery systems, enhancing the solubility and stability of therapeutic agents.
- Food Industry Applications: It can be used in food chemistry for flavoring and as a stabilizing agent, contributing to the development of new food products with enhanced taste and shelf life.
Documents
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
Product Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
*Catalog Number
*Lot Number
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.
*Catalog Number
*Lot Number
Safety Data Sheets (SDS)
The SDS provides comprehensive safety information on handling, storage, and disposal of the product.
DownloadProduct Specification (PS)
The PS provides a comprehensive breakdown of the product’s properties, including chemical composition, physical state, purity, and storage requirements. It also details acceptable quality ranges and the product's intended applications.
DownloadCertificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
*Catalog Number
*Lot Number
Certificates Of Origin (COO)
This COO confirms the country where the product was manufactured, and also details the materials and components used in it and whether it is derived from natural, synthetic, or other specific sources. This certificate may be required for customs, trade, and regulatory compliance.